Помідор. моделі захворювань
Фітофтороз
Фітофтороз томатів, спричинений грибком Phytophthora infestans, є однією з найбільш руйнівних хвороб рослин. У Європі вона призвела до голоду та еміграції. Це одна з найважливіших хвороб, і тому для неї існує безліч моделей. P. infestans облігатний паразит. Він може жити лише в зеленій тканині своїх господарів. Економічно важливими рослинами-хазяїнами є картопля, томати та баклажани. У прохолодному кліматі взимку патоген не знаходить зеленої тканини і змушений зимувати в інфікованих бульбах або в плодових тілах - ооспорах. Ооспори утворюються лише там, де два різних типи спарювання P. infestans присутні. Про це повідомляється в Європі протягом останніх 25 років. Ще більшу роль відіграє сплячка в інфікованих бульбах, залишених на полі в якості добровольців через малий розмір або з інших причин, а також відходів, що залишилися на полі після картоплесховища.
Новітні лабораторні методи дозволили нам перевірити наявність латентно інфікованих бульб у насінні картоплі. Це показало, що ми повинні очікувати цього в насінні картоплі. Кількість латентно інфікованого насіння залежить від епідемії фітофторозу в минулому сезоні в регіоні, де виробляється насіння.
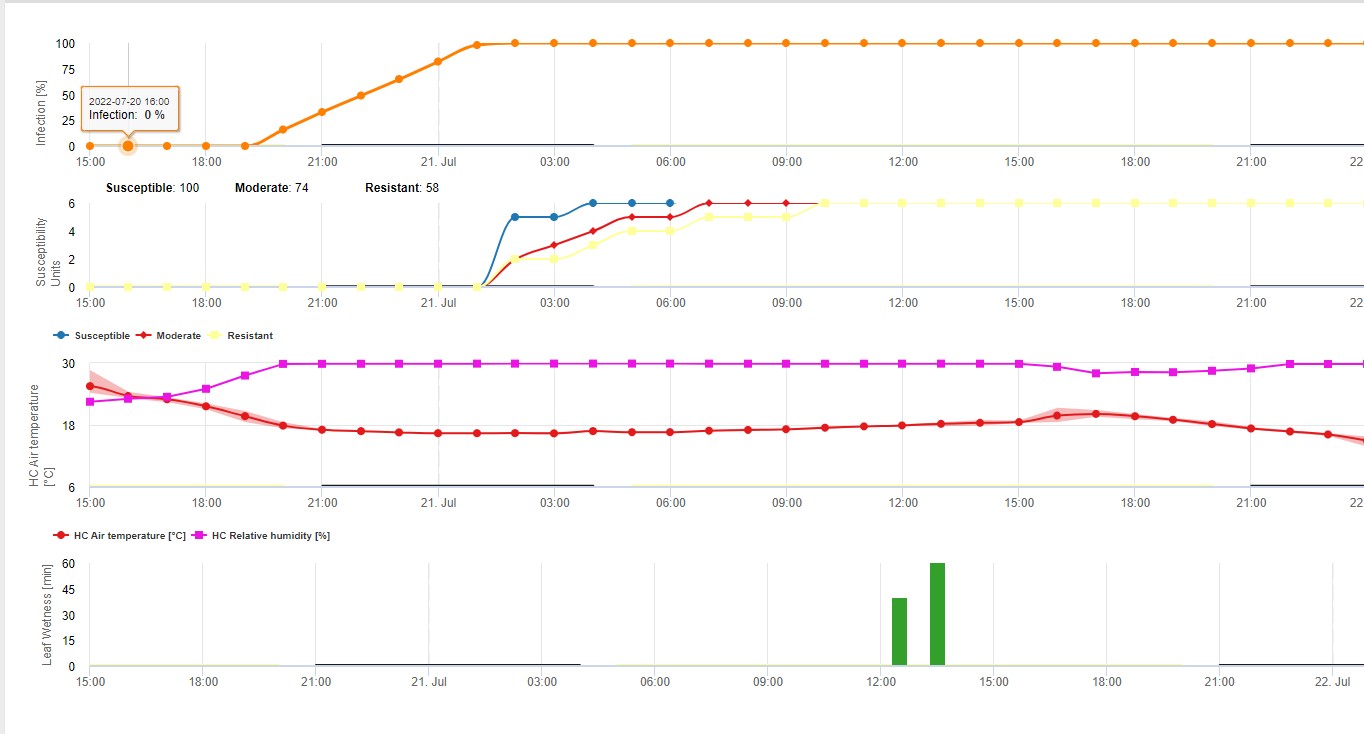
P. infestans Як і інші ооміцети, росте в міжклітинному просторі своїх господарів. Системний ріст підсилюється високою відносною вологістю та високим вмістом води в ґрунті або низьким вмістом кисню в ґрунті. Рослини, утворені латентно або симптоматично інфікованими бульбами, демонструють тривалий системний ріст у періоди перезволоження ґрунту. Вранці під час і після таких періодів ви побачите паростки картоплі, вкриті білими спорангіями. Спорангії в ооміцетів утворюються за відсутності світла, якщо відносна вологість і температура достатньо високі. Для P. infestans Утворення спорангіїв відбуватиметься в ночі з відносною вологістю вище 90% і температурою вище 10°C. Спорангії можуть поширюватися дощем або вітром.
У літературі можна знайти інформацію про те, що спорангії проростають і заражають подібно до конідій. Спорангії в ооміцетів зазвичай проростають зооспорами, які є рухливими у вільній воді. Зооспори пливуть до стоми, через яку вони заражають хазяїна. Джим Дікон з Інституту клітинної та молекулярної біології Единбурзького університету виявив, що при температурі 12°C і нижче більшість спорангіїв вивільняє зооспори, тоді як при температурі вище 20°C більшість спорангіїв проростає як конідії з зародковими трубками. Тому зараження P. infestans в прохолодному кліматі, найімовірніше, обмежується наявністю вільної вологи, яку може давати роса в ночі, коли відносна вологість повітря перевищує 90%, необхідну для утворення спорангіїв. Більш сильне зараження слід очікувати під час дощу, який поширює зооспори по картопляному полю і призводить до експоненціального зростання кількості інфікованих рослин.
У сильно заражених рослинах патоген системно проникає в усі органи рослини, включаючи бульби. У ситуаціях з сильним тиском хвороби листя картоплі необхідно знищити гербіцидом, щоб уникнути інфікування бульб.
У FieldClimate реалізовані моделі NOBLIGHT і FRY
Модель МІП
Модель IPI показує негативний прогноз, який був розроблений BUGIANI, CAVANNI, I. PONTI для регіону Емілія-Романья в Італії. Вона використовується для оцінки дати першого обприскування проти цієї хвороби на томатах.
Опис моделі: Ця модель генерує індекси інфекційного потенціалу (ІПІ), які прогнозують найбільш ймовірне збільшення кількості посівного матеріалу Фітофтора infestans у навколишньому середовищі. В Італії IP-індекси використовуються разом з рослинами-індикаторами та пастками для спор, щоб попередити фермерів про те, коли потрібно починати обприскування. Модель не дає рекомендацій щодо подальшого застосування фунгіцидів. Негативний прогноз не може бути використаний на територіях з постійним вирощуванням сільськогосподарських культур, а також на територіях, що не зимують.
Функціональність: Для розрахунку добового IPI відносні індекси середньої температури, відносної вологості та опадів розраховуються незалежно один від одного і комбінуються шляхом множення індексу температури на індекс опадів або на індекс відносної вологості. Кумулятивний щоденний IPI за певний період використовується моделлю для оцінки ризику фітофторозу. IPI-модель використовується як негативний прогноз в Італії. Коли значення IPI перевищує 15, починають обприскувати томати проти фітофторозу. Поки він залишається нижчим, обприскування не показане.
Результат: FieldClimate відображає постійно зростаюче значення IPI-значення. µLink відображає лінію, що постійно зростає. Коли IPI-значення досягає 18, внизу графіка відображається якісна лінія.
FieldClimate зупиняє розрахунок, якщо температура залишається нижче 11°C протягом 96 годин. Він починає розрахунок знову, якщо температура не опускається нижче 6°C протягом 96 годин. Максимальне значення цього розрахунку - 40. Для розрахунку щоденного IPI відносні індекси середньої температури, відносної вологості та опадів розраховуються незалежно і комбінуються шляхом множення індексу температури на індекс опадів або на індекс відносної вологості. IPI потрібно розраховувати лише тоді, коли дні мають мінімальну температуру вище 7°C, середню температуру від 9°C до 25°C і більше 0,2 мм опадів або середню відносну вологість більше 80%. Сприятливі погодні умови для Phytophthora infestans призводять до позитивного значення IPI. Функції для розрахунку індексу наведені на графіках поруч.
Модель IPI для фітофторозу томатів є моделлю негативного прогнозу. Вона корисна лише в тих регіонах, де немає постійного вирощування томатів. Це означає, що в районах з морозами взимку. На таких територіях інокуляційний потенціал Phytophtora infestans зменшується протягом зими і повинен знову нарощуватися навесні. Модель IPI вказує на накопичення посівного матеріалу в полі. Якщо значення IPI досягає 15, то перше обприскування призначається на ділянці, для якої була розроблена модель. Якщо ви використовуєте ділянку в іншій області, будь ласка, перевірте, чи дійсне це значення для вас.
Негативний прогноз
Використання негативного прогнозу означає відмову від обприскування до тих пір, поки прогноз відповідає на питання про наявність патогена в полі з NO. Це пояснює термін "негативний прогноз". Негативний прогноз Шредтера і Ульріха був опублікований в 1972 році. Він використовує температуру, вологість листя або високу відносну вологість і дощ для оцінки поширення патогена в полі. Значення в діапазоні від 0 до 400 вказує на поширення P. infestans в польових умовах. Це значення збільшується, якщо температура повітря знаходиться в діапазоні від 15°C до 20°C, якщо відносна вологість вища за 70%. Воно збільшується швидше, якщо відносна вологість вища за 90% і є опади або якщо вологість листя перевищує 4 години. Якщо така ситуація триває довше, ніж 10 годин, збільшення відбувається швидше.
Пороги: Шредтер і Ульріх визначають значення 150 як таке, що відповідає захворюваності в області 0,1%. Значення 250 відповідає захворюваності 1%. Вони припускають, що після року з низьким тиском фітофторозу в зоні виробництва насіння немає необхідності в обприскуванні до досягнення значення 250. Якщо передбачається більша кількість посівного матеріалу, обприскування слід починати з 150.
Тоді як оригінальна модель визначає початок розрахунку з появою сходів на конкретному полі, ми змінили початок розрахунку на правило, що базується на температурі, щоб переконатися, що ми розраховуємо, як тільки з'являться перші можливі помідори. Ми будемо розраховувати, як тільки температура з 10:00 до 18:00 буде вищою за 8°C, а нічна температура ніколи не буде нижчою за 2°C.
Негативний прогноз дуже успішно використовувався з 1972 року до дев'яностих років минулого століття. Це був час, коли ми не змогли знайти резистентність до металаксилу. Перше обприскування в ці роки зазвичай проводилося металаксилом, і за допомогою нього поле можна було очистити від P. infestans. Зараз великі площі мають стійкість до цієї сполуки, і ми не маємо жодного фунгіциду, який би демонстрував подібний очищувальний ефект.
На ділянках, де картопля під плівкою вирощується поряд з картоплею у відкритому ґрунті, ми рекомендуємо починати обприскування, як тільки з картоплі під плівкою буде знято поліетиленову плівку. Хвороба може розвиватися під плівкою, і після зняття плівки вкрита картопля стане джерелом збудника хвороби.
P. infestans росте системно всередині паростка. Це важливо, якщо ми маємо латентно інфіковане насіння. Системний ріст значно сприятливіший у перенасиченому водою ґрунті. Для отримання інформації про насиченість ґрунту водою ми рекомендуємо використовувати Датчики водяних знаків. Водяні знаки дуже економічні і дуже корисні для поливу картоплі. Якщо протягом декількох годин після появи сходів тиск води на датчику водяного знаку нижче 10 сБар (100 мБар) і температура повітря вище 10°C, ми повинні припустити, що існують сприятливі умови для системного росту патогена, і ми повинні почати з обприскування проти фітофторозу. Модель NoBlight: Значення ступеня ураження визначаються відповідно до моделі штату Мейн (Автор: Стівен Б. Джонсон, доктор філософії, спеціаліст із сільськогосподарських культур).
Джерело: http://umaine.edu/publications/2418e/#table
Модель FRY
W.E.FRY (1983) опублікував свою роботу, присвячену інфікуванню картоплі з різним рівнем сприйнятливості при різних тривалостях відносної вологості вище 90% або вологості листя і температурі. На основі цих результатів він розробив модель зараження картоплі фітофторозом, а на наступному етапі - модель для оцінки інтервалу між обприскуваннями фунгіцидом клорантонілом (Браво).
Сприйнятливі сорти можуть бути інфіковані протягом коротших вологих періодів, і тяжкість хвороби буде вищою. Тоді як помірно сприйнятливі та стійкі сорти потребують більш тривалого вологого періоду або більш високих температур для зараження, а тяжкість хвороби буде меншою.
Для сприйнятливих сортів максимальна оцінка інфекційного періоду може становити 7 балів, тоді як для помірно сприйнятливих сортів - 6, а для стійких - лише 5 балів. Таким же чином оцінка інтервалу між обприскуваннями знову ж таки залежить від рівня сприйнятливості сорту. Обприскування необхідне, якщо до останнього обприскування пройшло більше 6 днів, а кількість накопичених одиниць фітофторозу перевищує норму: 30 для сприйнятливих сортів, 35 для помірно сприйнятливих сортів і 40 для помірно стійких сортів.
Ця модель дуже корисна для оцінки необхідності нового обприскування. Ми можемо почати накопичувати одиниці мальків з дати останнього обприскування. Якщо накопичене значення перевищує поріг, нам доведеться обприскувати знову. Fry, WE, AE Apple & JA Bruhn (1983). Оцінка прогнозів фітофторозу картоплі, модифікованих для врахування стійкості хазяїна та вивітрювання фунгіцидів. Фітопатологія 73:1054-1059.
Ранній фітофтороз
Ранній фітофтороз картоплі та томатів
Рендалл К. Роу, Саллі А. Міллер, Річард М. Рідел, Дорадча служба Університету штату Огайо
Фітофтороз є дуже поширеним захворюванням як картоплі, так і томатів. Вона викликає плямистість листя і бульб у картоплі, а у томатів - плямистість листя, плодову гниль і ураження стебла. Захворювання може виникати в широкому діапазоні кліматичних умов і може бути дуже руйнівним, якщо його не контролювати, часто призводячи до повної дефоліації рослин. Всупереч назві, вона рідко розвивається на ранніх стадіях, але зазвичай з'являється на зрілому листі.
Симптоми
На листках обох культур перші симптоми зазвичай з'являються на старих листках і являють собою невеликі, неправильної форми, від темно-коричневого до чорного кольору, мертві плями розміром від крапки до 1/2 дюйма в діаметрі. Коли плями збільшуються, можуть утворюватися концентричні кільця внаслідок нерегулярного росту організму в тканині листка. Це надає ураженню характерного вигляду "плями-мішені" або "яблучне око". Навколо кожної плями часто є вузький жовтий ореол, а місця ураження, як правило, облямовані жилками. Коли плям багато, вони можуть зростатися, що призводить до пожовтіння та відмирання заражених листків. Зазвичай першими заражаються найстаріші листки, які засихають і опадають з рослини в міру того, як хвороба прогресує вгору по головному стеблу.
На томатах стеблові інфекції можуть виникати в будь-якому віці, що призводить до появи невеликих темних, злегка заглиблених ділянок, які збільшуються, утворюючи круглі або витягнуті плями зі світлішими центрами. На стеблах часто утворюються концентричні плями, схожі на ті, що на листках. Якщо для пересадки томатів використовується заражене насіння, розсада може засохнути незабаром після появи сходів. Коли на стеблах пересаджених рослин або розсади розвиваються великі ураження на рівні ґрунту, рослини можуть бути опоясані - стан, відомий як "шийна гниль". Такі рослини можуть загинути під час висадки в поле або, якщо стебла ослаблені, зламатися на початку сезону. Деякі рослини можуть вижити зі зменшеною кореневою системою, якщо частини стебел над раком розвинуть коріння в місцях контакту з ґрунтом. Такі рослини, однак, зазвичай дають мало плодів або взагалі не дають плодів. Ураження стебла набагато рідше зустрічається на картоплі і є руйнівним для неї.
Коли рання гниль вражає томати на стадії цвітіння, може спостерігатися опадання цвіту і плямистість плодових стебел, а також втрата молодих плодів. На старих плодах фітофтороз викликає темні, шкірясті вдавлені плями, зазвичай в місці прикріплення плодоніжки. Ці плями можуть збільшуватися і охоплювати всю верхню частину плоду, часто показуючи концентричні позначки, подібні до тих, що є на листі. Уражені ділянки можуть бути вкриті оксамитовою чорною масою спор. Плоди також можуть бути інфіковані на зеленій або стиглій стадії через ростові тріщини та інші рани. Заражені плоди часто опадають, не досягнувши зрілості.
На бульбах картоплі рання фітофтороза призводить до поверхневих уражень, які виглядають трохи темнішими, ніж прилегла здорова шкіра. Ураження, як правило, злегка заглиблені, круглі або неправильної форми і варіюються в розмірах до 3/4 дюйма в діаметрі. Зазвичай між здоровою та ураженою тканиною є чітко окреслена, а іноді трохи піднята межа. Зсередини тканина має коричневу або чорну коркову суху гниль, зазвичай глибиною не більше 1/4 - 3/8 дюйма. На старих ураженнях можуть утворюватися глибокі тріщини. Бульбова інфекція в умовах Огайо зустрічається рідко.
Збудник
Ранній фітофтороз викликається грибком Alternaria solani, який виживає в інфікованих тканинах листя або стебла на ґрунті або в ґрунті. Цей грибок повсюдно присутній на полях, де вирощують ці культури. Він також може переноситися на насінні томатів та бульбах картоплі. Спори утворюються на заражених рослинних рештках на поверхні ґрунту або на активних уражених ділянках у досить широкому діапазоні температур, особливо в умовах чергування вологих і сухих умов. Вони легко переносяться повітряними потоками, вітром, дощовими бризками та поливною водою. Зараження сприйнятливих тканин листя або стебла відбувається в теплу, вологу погоду з рясними росами або дощами. Ранній фітофтороз може розвиватися досить швидко в середині та наприкінці сезону і є більш серйозним, коли рослини перебувають у стресовому стані через погане живлення, посуху або інших шкідників. Зараження бульб картоплі відбувається через природні отвори на шкірці або через пошкодження. Бульби можуть контактувати зі спорами під час збирання врожаю, і ураження може продовжувати розвиватися під час зберігання.
Передумови
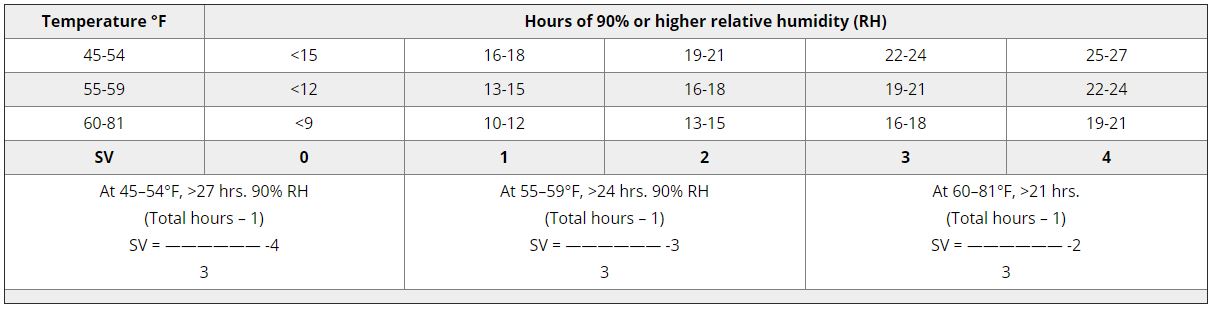
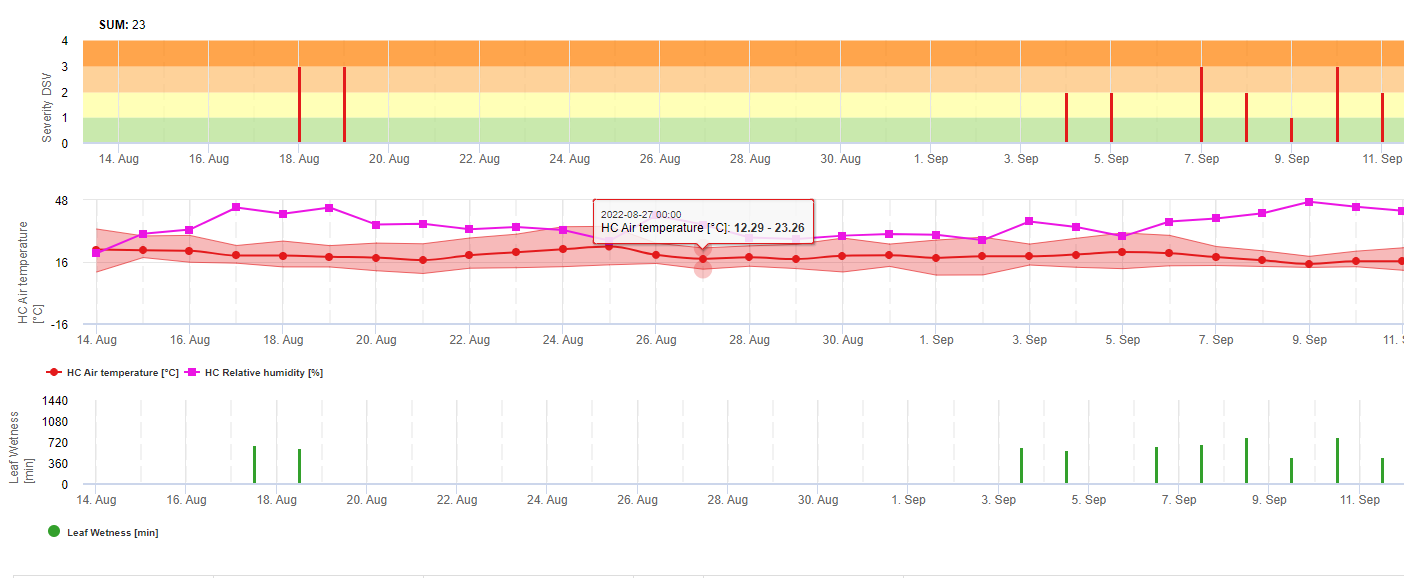
TOMCAST (TOMato disease foreCASTing) - це комп'ютерна модель, заснована на польових даних, яка намагається передбачити розвиток грибкових захворювань, а саме раннього фітофторозу, септоріозу листя та антракнозу на томатах. Реєстратори даних, розміщені на полях, реєструють погодинні дані про вологість і температуру листя. Ці дані аналізуються протягом 24 годин і можуть призвести до формування Значення тяжкості захворювання (DSV); по суті, це приріст розвитку хвороби. У міру накопичення DSV, тиск хвороб продовжує наростати на культуру. Коли кількість накопичених DSV перевищує інтервал між обприскуваннями, рекомендується застосування фунгіциду, щоб зменшити тиск хвороби.
TomCast
Час застосування фунгіцидів проти раннього фітофторозу, септоріозу листя та антракнозу
Система прогнозування хвороб на основі погодних умов під назвою TOMCAST, розроблена доктором Роном Пітбладо в Ріджтаунському коледжі сільськогосподарських технологій в Онтаріо, Канада, може бути використана для визначення часу застосування фунгіцидів проти трьох грибкових захворювань: раннього фітофторозу (викликається Alternaria solani), септоріозної плямистості листя (викликається Septoria lycopersici) та антракнозу плодових (викликається Colletotrichum coccodes). Якщо фітофтороз присутній у вашому або сусідніх округах, або існують умови для переміщення спор на вашу територію, використовуйте систему прогнозування фітофторозу Simcast, щоб визначити час застосування фунгіцидів.
ВАЖЛИВЕ ЗАСТЕРЕЖЕННЯ:
ТОМКАСТ не є корисним на фермах, які мають історію бактеріальних захворювань. Якщо у вас часто виникають проблеми з бактеріальною плямистістю, плямистістю або раком на томатах, вам не слід використовувати ТОМКАСТ, оскільки рекомендовані інтервали між обприскуваннями будуть недостатніми для контролю бактеріальних захворювань, якщо ви змішуєте мідь з фунгіцидами в баках. Знайдіть інформацію про зменшення бактеріальних захворювань тут: http://extension.psu.edu/plants/vegetable-fruit/news/2015/farming-like-you-expect-bacterial-diseases
TOMCAST використовує дані про вологість і температуру листя для розрахунку показників тяжкості хвороби (DSV), як показано в Таблиці 1.
TOMCAST походить від оригінальної моделі F.A.S.T. (Forecasting Alternaria solani on Tomatoes), розробленої докторами Медденом, Пенніпекером і Макнабом в Університеті штату Пенсильванія (PSU). Модель PSU F.A.S.T. була в подальшому модифікована доктором Пітбладо в коледжі Ріджтаун в Онтаріо в те, що ми зараз визнаємо як модель TOMCAST, яка використовується в Університеті штату Огайо.
DSV Значення тяжкості хвороби (DSV) - це одиниця виміру, що відповідає певному інкременту розвитку хвороби (раннього фітофторозу). Іншими словами, DSV - це числове представлення того, наскільки швидко або повільно хвороба (ранній фітофтороз) накопичується на томатному полі. DSV визначається двома факторами: вологістю листя і температурою в години "мокрого листя". Зі збільшенням кількості годин з мокрим листям і температури DSV накопичується швидше. Нижче наведено діаграму "Тяжкість хвороби".
І навпаки, за меншої кількості годин зволоження листя та нижчої температури DSV накопичуються повільніше, якщо взагалі накопичуються. Коли загальна кількість накопичених DSV перевищує встановлену межу, яка називається інтервалом або порогом обприскування, рекомендується обприскування фунгіцидом, щоб захистити листя і плоди від розвитку хвороб.
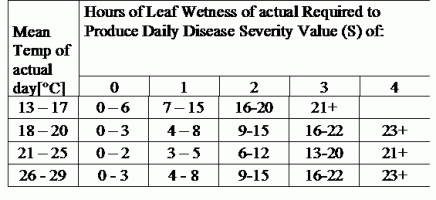

Інтервал між обприскуваннями (який визначає, коли слід обприскувати) може коливатися в межах 15-20 DSV. Точне значення DSV, яке повинен використовувати виробник, зазвичай надається переробником і залежить від якості плодів і кінцевого використання томатів. Дотримання інтервалу обприскування 15 DSV є консервативним використанням системи TOMCAST, тобто ви будете обприскувати частіше, ніж фермер, який використовує інтервал обприскування 19 DSV з системою TOMCAST. Компроміс полягає в кількості обприскувань протягом сезону і потенційній різниці в якості плодів.
ЗА ДОПОМОГОЮ TOMCAST: Томати, вирощені в радіусі 10 миль від станції спостереження, можуть скористатися функцією управління хворобами TOMCAST для прогнозування раннього фітофторозу, септоріозу та антракнозу. Якщо ви вирішите спробувати TOMCAST в цьому сезоні, будь ласка, майте на увазі три дуже важливі концепції.
Перше: Якщо ви вперше використовуєте систему, рекомендується внести в програму лише частину ваших посівних площ, щоб побачити, наскільки вона відповідає вашим стандартам якості та стилю роботи.
Друге: використовуйте TOMCAST як орієнтир для вибору часу внесення фунгіцидів, розуміючи, що в деякі сезони ви можете внести більше продукту, ніж може вимагати програма з встановленим графіком.
Третє: Чим далі томатне поле знаходиться від місця збору даних, тим більша ймовірність викривлення в накопиченні DSV, тобто звітне значення може бути на кілька DSV вищим або нижчим від того, що спостерігається на місці розташування поля. Це слід враховувати, коли застосування фунгіцидів, ймовірно, відбудеться через кілька днів. Прослухайте звіти про DSV сусідніх станцій і проведіть тріангуляцію до свого місця розташування - це найкращий спосіб приблизно оцінити накопичення DSV.
ПЕРШЕ ОБПРИСКУВАННЯ З ВИКОРИСТАННЯМ TOMCAST:Протягом багатьох років точилися дискусії щодо застосування першого обприскування при використанні ТОМКАСТ. Правило, викладене в Посібнику з овочівництва 1997 року, зосереджується на даті посадки.
Рослини томатів, висаджені в поле до 20 травня, слід обприскувати, коли DSV для цієї зони перевищує 25 або коли настає безпечна дата - 15 червня. Безпечна дата використовується тільки в тому випадку, якщо ви не проводили обприскування з 20 травня, і є засобом для усунення початкового зародка хвороби. Після першого обприскування ці томати обробляють, коли перевищується обраний інтервал обприскування (діапазон 15-20 DSV).
Томати, висаджені після 20 травня, обробляють, коли вони перевищують обраний інтервал обприскування (діапазон 15-20 DSV) або коли вони не були оброблені до безпечної дати 15 червня. Тому дуже важливо порівняти дату посадки томатів з датою початку звітності по DSV на цій ділянці, щоб керуватися нею в процесі прийняття рішення про обприскування.
Борошниста роса
Борошниста роса томатів може бути викликана трьома збудниками у всьому світі.
Leveillula taurica (Oidiopsis taurica) є патогеном широкого спектру видів хазяїв у теплому посушливому та напівпосушливому кліматі в Азії, Середземномор'ї, Африці, а віднедавна і на південному заході Сполучених Штатів Америки.
Erysiphe orontii (E. cichoracearum і E. polyphaga) - ще один вид, поширений серед багатьох рослин-господарів як у помірних, так і в тропічних регіонах.
І як третій вид Odium lycopersicum.
Фактори розвитку захворювання:
- рівень відносної вологості > 50% (оптимальний RH > 90%)
- вільна вода на поверхні листя не потрібна
- температурний діапазон: 10-35 °C (найкраще нижче 30 °C)
Борошниста роса - це хвороба, що спричиняється інокулятом. Тому можна визначити лише ризиковані періоди, основним фактором ураження, наприклад, спалаху, є початковий інокулят (який активний у широкому діапазоні температур). Отже, для стратегій контролю: поєднуйте моделювання ризикованого періоду з моніторингом грибкового інокуляту (хвороби) в полі!
Томат відкритого ґрунту
Сіра пліснява
Сіра пліснява (Botrytis cinerea) зимує у вигляді склероціїв або міцелію в рослинних рештках і може переноситися з насінням у вигляді спор або міцелію на деяких культурах. Інші культури також можуть слугувати джерелом збудника і, ймовірно, перехресно інфікувати. Конідії переносяться повітряно-крапельним шляхом, а також на поверхні дощових крапель. Висока відносна вологість повітря необхідна для інтенсивного спороношення. У польових умовах спори, що потрапляють на рослини томатів, проростають і викликають інфекцію, коли на поверхню рослини потрапляє вільна вода від дощу, роси, туману або поливу.
Оптимальна температура для інфікування становить від 18° до 24° C, і зараження може відбутися протягом 5 годин. Високі температури, вище 28°С, пригнічують ріст і утворення спор. Сприятливим місцем для зараження є в'янучі квіти, але інфекція може також виникнути в результаті прямого контакту з вологим зараженим ґрунтом або рослинними рештками. У теплиці ураження стебла розвивається або шляхом прямої колонізації ран, або через інфіковане листя. Присутність зовнішніх поживних речовин, таких як пилкові зерна в інфекційній краплі, може значно посилити інфекцію. Вважається, що тип рани впливає на розвиток ураження стебла; повідомляється, що обривання листя призводить до меншої частоти ураження стебла, ніж обрізання листя ножем із залишенням пенька.
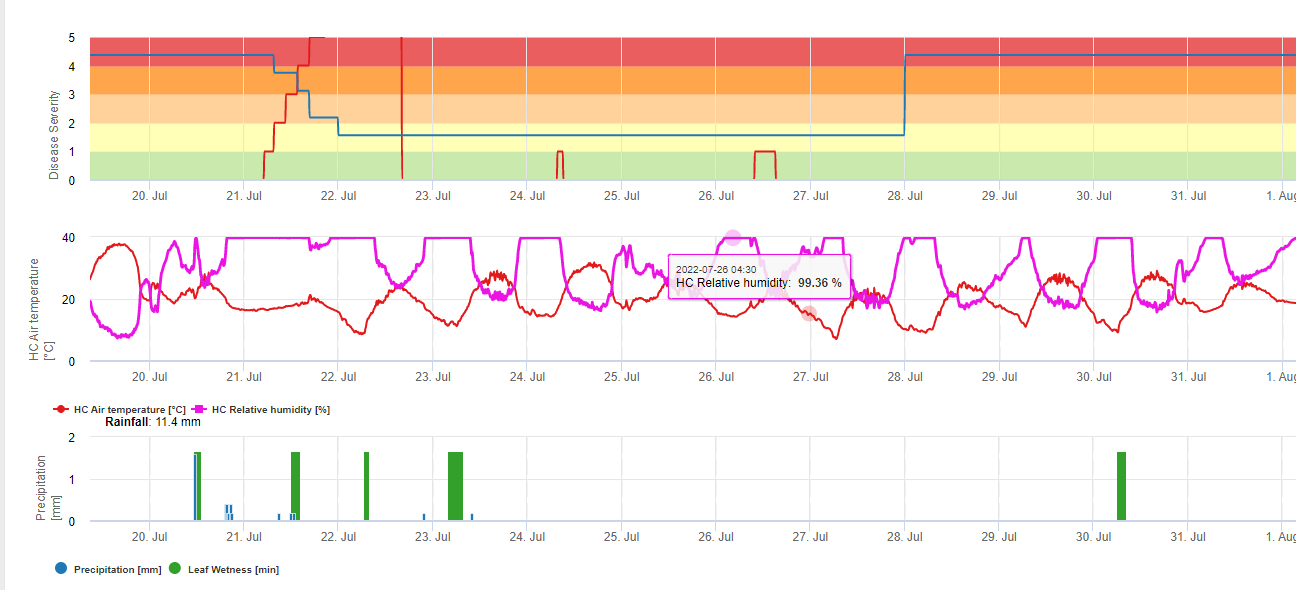
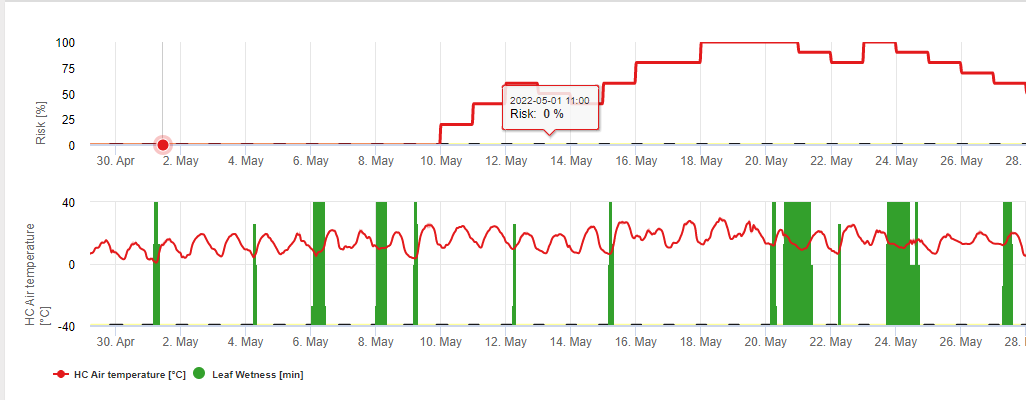
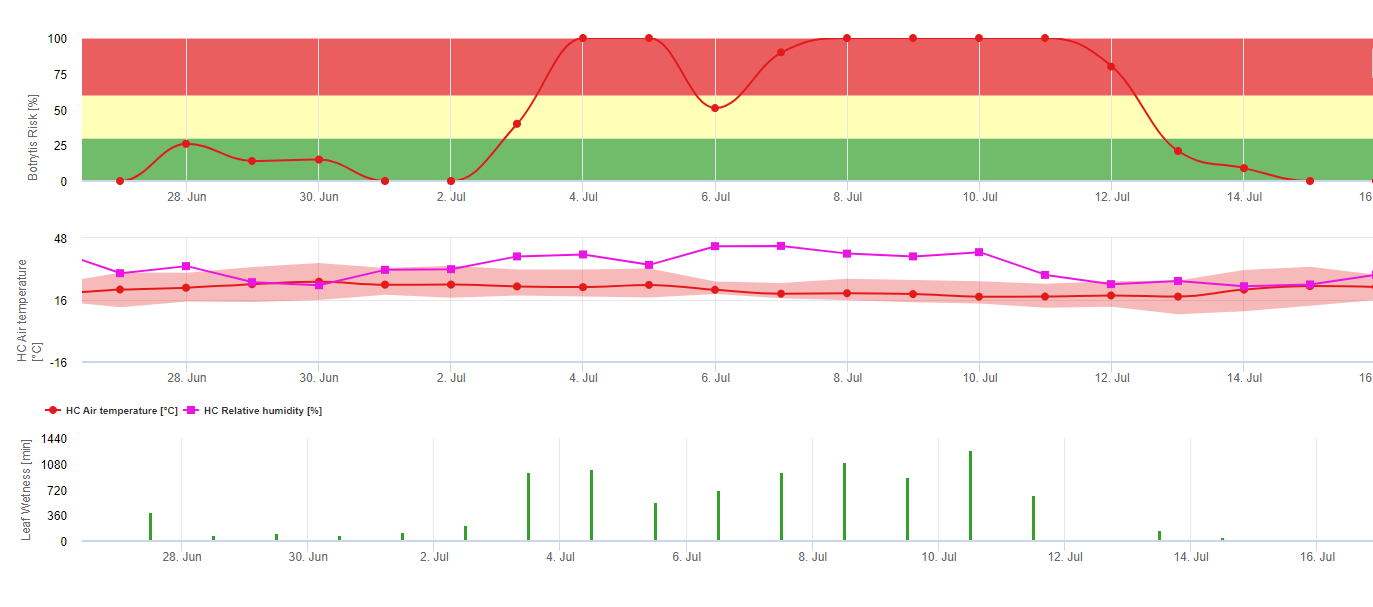
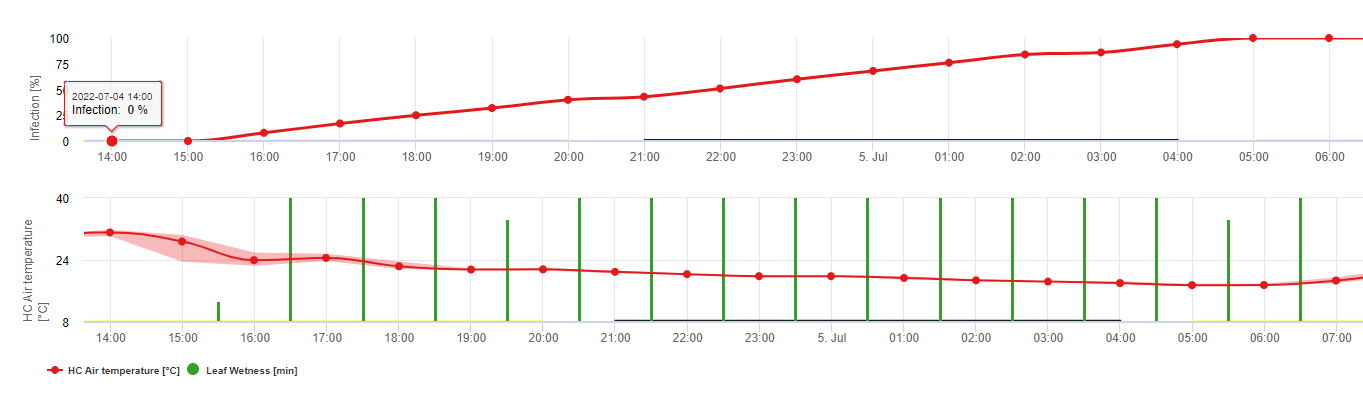
FieldClimate вказує на ризик виникнення Botrytis cinerea інфекції на основі періодів вологості листя та температури. На графіку нижче показано тривалість періоду мокрого листя в залежності від фактичної температури, необхідної для розвитку інфекції Botrytis. Якщо ризик вищий за 0, кожен період вологості листя довший за 4 години збільшує ризик у тому ж співвідношенні. День з періодом вологості листя менше 4 годин вважається сухим днем і зменшує ризик на 20% від фактичного значення.
На графіку показано кореляцію між тривалістю зволоження листя та температурою, що призводить до ризику виникнення 30% B.cinerea інфекція.
FieldClimate: Ботритис ризик, розрахований на основі температури та періоду вологості листя, виміряних на станції. FieldClimate Ботритис Модель ризику дає значення ризику від 0 до 100%. Це значення вказує на тиск B. cinerea на той час. Якщо ми маємо значення 100%it, це означає, що кілька разів був період вологості, достатній для інфікування сприйнятливої тканини (ми обчислюємо так звані "вологі точки" (масив між вологістю листя, температурою з максимумом спочатку 38400 точок (початок сезону, який відображає ризик 30%). Після цього періоду кожен вологий період з приблизно 4000 вологих точок (масив) збільшує ризик до 10% або, з іншого боку, кожен сухий період зменшує ризик на 1/5 від попереднього значення. Заявка проти B. cinerea залежить від фруктів та виробничих цілей.
Плямистість листя
Смс від..: T. А. Зіттер, кафедра патології рослин, Корнельський університет ; Інформаційний бюлетень Стр: 735.80 Дата: 12-1987.
Септоріоз листя томатів, спричинений грибком Septoria lycopersici зустрічається на томатах по всьому світу. Грибок вражає лише пасльонові рослини, серед яких томат є найважливішим. Томати часто можуть бути заражені одночасно плямистістю листя і раннім фітофторозом (Altemaria solani), але ці дві хвороби можна легко відрізнити, а заходи боротьби з ними схожі.
Симптоми
Септоріоз листя може виникнути на будь-якій стадії розвитку рослини. Симптоми можуть з'являтися на молодих тепличних саджанцях, готових до пересадки, або вперше спостерігатися на нижніх, старіших листках і стеблах під час зав'язування плодів. Час появи симптомів може бути пов'язаний з джерелом посівного матеріалу та факторами навколишнього середовища, що буде розглянуто пізніше. Невеликі, змочені водою круглі плями діаметром від 1,6 до 3,2 мм спочатку з'являються на нижньому боці старих листків. Центри плям сірі або коричневі, а самі плями мають темно-коричневий край. Коли плями дозрівають, вони збільшуються до 6,4 мм в діаметрі і можуть зливатися. У центрі плям є багато темно-коричневих, схожих на прищі структур, які називаються пікнідами - плодовими тілами гриба. Ці структури досить великі, щоб їх можна було побачити неозброєним оком або за допомогою ручної лупи. Пікніди відсутні на уражених раннім фітофторозом рослинах і на уражених сірою гниллю листків, Stemphylium solaniяка поширена в регіонах зі стабільно теплими та вологими умовами. Септоріозна плямистість листя також не має мішенеподібних уражень, характерних для Фітофтороз альтемарії. Плями також можуть з'являтися на стеблах, чашечках і квітках, але рідко на плодах. Сильно заражене листя жовтіє, засихає і опадає. Таке опадання призводить до сонячних опіків плодів.
Епідеміологія
У вологих умовах у пікнідах утворюються численні спори (конідії), які виділяються, коли плодові тіла дозрівають. Температурний діапазон для спороношення варіює від 15° до 27°C, оптимальною є температура 25°C. Спори можуть поширюватися вітром, дощовими бризками, руками та одягом збирачів, комахами, такими як жуки, та культиваційним обладнанням. Після поширення спори можуть проростати протягом 48 годин у вологих умовах і при сприятливій температурі. Плями на листках можуть з'явитися протягом 5 днів, пікніди - протягом 7-10 днів, а спороношення повторюється протягом 10-13 днів. Оскільки для зараження спорами через продихи необхідна вільна волога, тривала роса і дощові дні (100% відносна вологість повітря протягом 48 годин, накопичена протягом декількох днів) сприяють розвитку хвороби. Хоча гриб не є мешканцем ґрунту, він може зберігатися з сезону в сезон на залишках хворих рослин, що потрапили в ґрунт (найчастіше в полі, але іноді і в теплиці). Патоген також може зимувати на пасльонових бур'янах. До чутливих бур'янів належать: берізка польова (Datura stramonium), однорічник, що розмножується насінням хрінниця (Solanum carolinense), багаторічник, що розмножується насінням і кореневищами; суниця гладка (Фізаліс підглабровий), багаторічна рослина, що розмножується кореневищами та насінням; та паслін чорний (Solanum nigrum), однорічна рослина, що розмножується насінням. Крім томатів, до інфікованих культур належать картопля та баклажани. Доведено, що насіння томатів може переносити спори і давати інфіковані сходи, але чи дійсно патоген передається через насіння, невідомо.
Антракноз плодових гнилей
Кілька видів рослинних патогенних грибів у роді Коллетотрихум (Colletotrichum) викликають антракноз перцю та багатьох інших овочів і фруктів. До кінця 1990-х років антракноз перцю і томатів був пов'язаний тільки зі стиглими або дозріваючими плодами. З того часу встановилася більш агресивна форма хвороби. Ця форма вражає перець на будь-якій стадії розвитку плодів і може загрожувати прибутковості посівів перцю в районах, де вона закріпилася. Ця хвороба також може вражати помідори, полуницю і, можливо, інші фруктові та овочеві культури.
Симптоми
Кругові або кутові вдавлені ураження розвиваються на незрілих плодах будь-якого розміру. Часто на окремих плодах утворюються множинні ураження. При важкому перебігу хвороби ураження можуть зливатися. Часто на поверхні уражених плодів утворюються концентричні кільця спор гриба від рожевого до помаранчевого кольору. У застарілих ураженнях можна спостерігати чорні структури, які називаються ацервули. Неозброєним оком вони виглядають як маленькі чорні крапки, а під мікроскопом - як пучки крихітних чорних волосків. Патоген швидко і рясно утворює спори і може швидко поширюватися по всій культурі перцю, що призводить до втрати врожаю до 100%. Ураження також може проявлятися на стеблах і листках у вигляді коричневих плям неправильної форми з темно-коричневими краями.
Збудник
Ця форма антракнозу перцю викликається грибком Colletotrichum acutatum. Патоген виживає на рослинних рештках заражених культур та на інших сприйнятливих видах рослин. Гриб не переноситься ґрунтом протягом тривалого часу за відсутності заражених рослинних решток. Гриб також може потрапляти в культуру з зараженим насінням. У теплі та вологі періоди спори переносяться з дощем або зрошувальною водою з хворих плодів на здорові. Хворі плоди слугують джерелом посівного матеріалу, що дозволяє хворобі поширюватися від рослини до рослини в межах поля. Грибок виживає в насінні та на ньому. Антракноз потрапляє в поле з інфікованими саджанцями або може виживати між сезонами в рослинних рештках чи на бур'янах-хазяїнах. Альтернативними хазяями є бур'яни та інші рослини родини пасльонових (томати, картопля, баклажани), хоча у Флориді інфікування цих рослин надзвичайно рідкісне. Плоди заражаються, коли спори гриба або заражені рештки потрапляють під час дощу на рослини перцю. Нові спори утворюються в зараженій тканині, а потім поширюються на інші плоди. Працівники також можуть переносити спори з обладнанням або під час роботи з зараженими рослинами. Зараження зазвичай відбувається в теплу, вологу погоду. Оптимальною температурою для розвитку хвороби є температура близько 80° F (27° C), хоча інфікування відбувається як за вищих, так і за нижчих температур. Серйозні втрати відбуваються під час дощової погоди, оскільки спори змиваються або розбризкуються на інші плоди, що призводить до збільшення кількості інфекцій. Хвороба частіше розвивається на зрілих плодах, які тривалий час перебувають на рослині, хоча вона може виникнути як на незрілих, так і на зрілих плодах. Антракноз може розвиватися при температурі від 15°C до 30°C. Але для розвитку інфекції необхідна тривала вологість листя. При оптимальній температурі від 20°C до 25°C потрібно ще 12 годин зволоження листя. Вища або холодніша температура потребує ще довшого періоду зволоження листя (для розрахунку не потрібна лінійна функція/масив). FieldClimate розраховує можливі події зараження на основі вологості листя та температури під час цієї події.
Листова пліснява
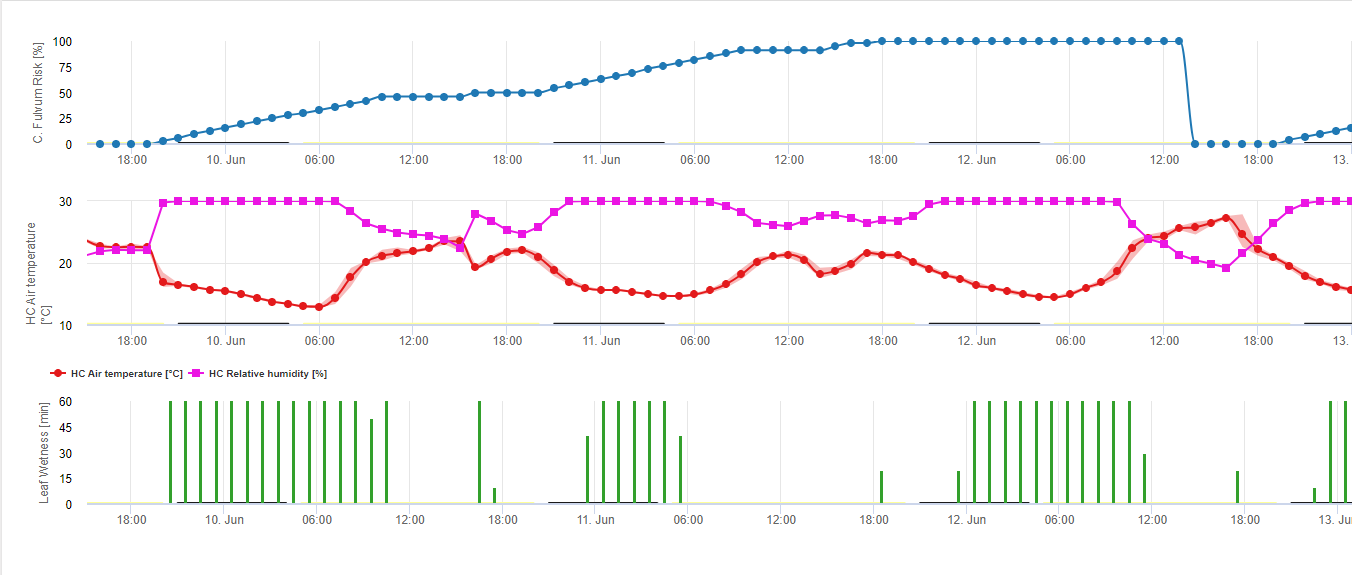
Пліснявіння листя, спричинене патогеном Fulvia fulva (Cladosporium fulvum) - це насамперед хвороба тепличних томатів, хоча вона може виникати і в польових умовах за прохолодної вологої погоди. Найбільш небезпечний у вологих тепличних умовах і в погано вентильованих пластикових будинках, грибок вражає рослини, вирощені в ґрунті, а також у гідропонному виробництві. Томат - єдина рослина, яка уражається цією хворобою. Фактичний взаємозв'язок між тяжкістю захворювання і втратою врожаю все ще не з'ясований. Однак в одному дослідженні було виявлено значне зниження врожайності через 6 тижнів після появи симптомів 50% на листі.
Патоген виживає на томатному полі:
- як сапрофіт на рослинних рештках або у вигляді конідій чи склероціїв у ґрунті;
- у вигляді конідій або спор (можуть виживати щонайменше один рік без хазяїна або за несприятливих умов);
- як забруднювач насіння.
Симптоми
Симптоми зазвичай проявляються лише на листі. Першими заражаються старіші листки, і грибок поступово поширюється вгору по рослині на молодші листки. Початкові симптоми на листках виглядають як блідо-жовті або зелені ділянки або плями з невизначеними краями. Вони часто спочатку з'являються на верхній поверхні листка. При сильному зараженні ці плями можуть зливатися, і тоді гине весь листок. Діагностичні симптоми з'являються на нижній поверхні листка, коли гриб спороношенням надає зараженій ділянці оливково-зеленого, оксамитового вигляду. Заражені листки з часом буріють, скручуються, в'януть і передчасно опадають. Дефоліація поступово прогресує вгору по рослині, коли грибок поширюється на молоде листя. Симптоми можуть іноді розвиватися на черешках, стеблах, квітконосах, квітах і плодах. Заражені квітки зазвичай гинуть до зав'язування плодів. Зелені та стиглі плоди можуть бути інфіковані і розвивати темну, шкірясту гниль на кінці стебла. Заражені плоди також можуть бути однобокими і мати почорнілі борозни.
Кліматичні умови для розвитку хвороби:
- рівні відносної вологості >85%
- вільна вода на поверхні листя
- оптимальна температура: 22-24 °C (проростання відбувається при 5-35 °C)
У FieldClimate ми визначаємо ризик виникнення Cladosporium fulvum зараження від вологості листя, відносної вологості та температури повітря. На графіку показано зараження томатів 22 квітня.
Фітофтороз
Фітофтора фітофтороз перцю викликається грибком Фітофторозна плямистість (Phytophthora capsici). Інші назви, що застосовуються до цієї хвороби перцю, - відсихання і фітофторозна коренева гниль, гниль крони, а також стеблова і плодова гниль. Всі ці назви можуть застосовуватися, оскільки уражаються всі частини рослини перцю. Захворювання призводить до серйозних втрат. Іншими культурами, ураженими хворобою, були баклажани, помідори, літні та зимові патисони і гарбуз. Також повідомлялося про огірок, кавун і диню медвяну. Патогеном, причетним до цих останніх культур, можуть бути Phytophthora parasitica або P. capsici. Фітофтороз перцю може вражати коріння, стебла, листя і плоди, залежно від того, на якій стадії рослини заражені. Фермер, який не знає, чого очікувати, може вперше зіткнутися з хворобою в середині сезону, коли рослини раптово в'януть і гинуть, коли вони досягають стадії плодоношення. Рано заражені рослини швидко гинуть, тоді як рослини, заражені пізніше, зазнають незворотного в'янення. Часто ці симптоми проявляються одночасно на кількох рослинах, розташованих у рядку або приблизно по колу. Заражені грибком саджанці висихають на рівні ґрунту, але відносно небагато рослин гине при прохолодних температурах. Набагато частіше хвороба вражає старіші рослини, які починають рано в'янути. Ураження стебла може відбуватися на рівні ґрунту та на будь-якому рівні стебла. Стебла знебарвлюються зсередини, руйнуються і з часом можуть дерев'яніти. Ураження може охоплювати стебло, що призводить до в'янення вище місця ураження, або ж рослини можуть в'янути і гинути через те, що гриб вражає верхні гілки до того, як ураження стебла стане достатньо сильним, щоб спричинити його руйнування.
P. capsici розмножується найшвидше в теплу і вологу погоду, утворюючи мільйони короткоживучих лимоноподібних спор на поверхні заражених рослин. Ці спори можуть розбризкуватися з ґрунту на рослини або між рослинами, а також переноситися водою, що тече по полю. Кожна особина може також випускати 20-40 рухливих спор, які можуть плавати на невеликі відстані у стоячій воді або насиченому вологою ґрунті до коріння рослин. Обидва ці види спор можуть утворюватися дуже швидко, і для цього потрібна наявність лише одного ізоляту P. capsici. Другий тип спор з набагато товстішими стінками утворюється всередині зараженої рослинної тканини і вимагає присутності щонайменше двох ізолятів P. capsici. Усе. P. capsici можна класифікувати як ізоляти A1 або A2, і ці товстостінні спори утворюються лише тоді, коли ізоляти A1 і A2 ростуть близько один до одного. Хоча ці спори утворюються повільніше, вони дуже важливі для життєвого циклу Фітофтора тому що вони можуть роками виживати в ґрунті, доки не буде висаджена сприйнятлива до них культура. З цієї причини, як тільки ці товстостінні спори потрапляють у ґрунт Фітофтора фітофтороз залишається. У FieldClimate ми розраховуємо утворення і зараження ооспорами (статевим шляхом) і утворення і зараження спорангіями (безстатевим шляхом). Для утворення ооспор необхідна наявність двох ізолятів P. capsiciмають товсті стінки всередині рослинної тканини і здатні зберігатися тривалий час, тоді як спорангії - це безстатева форма, яка швидко поширюється.
На листі спочатку з'являються невеликі темно-зелені плями, які збільшуються і стають білими, ніби ошпарені. Якщо інфіковані стебла рослини, відбувається незворотне в'янення листя. На заражених плодах спочатку з'являються темні, змочені водою плями, які покриваються білою пліснявою і спорами грибка. Плоди в'януть, але залишаються прикріпленими до рослини. Насіння зморщується і заражається грибком. Через широкий спектр хазяїв та різні фази, на яких рослини можуть бути заражені, зверніться до таблиці для уточнення уражених культур та Фітофтора задіяні види.
Симптоми очної гнилі томатів складаються з шкірястих коричневих плям, часто у вигляді концентричних кілець або смуг на зелених плодах. Поразки можуть з'являтися на плечі або, частіше, на кінчику квітки, де томат контактує з вологим ґрунтом. На патисонах (і на деяких інших культурах, перелічених у списку з симптомами на плодах) засмаглі або коричневі ураження можуть мати вигляд смуг або виглядати як великі круглі плями. У вологих умовах на поверхні з'являється білий ватоподібний міцелій і спори, а плоди, швидше за все, швидко загнивають від вторинних організмів.
Рекомендоване обладнання
Перевірте, який набір датчиків потрібен для моніторингу потенційних хвороб цієї культури.