Armut hastalık modelleri
Yağmur birikimi
Şiddetli yağmur, asmaların veya diğer bitkilerin yapraklarında kalan pestisitleri yıkayacaktır. 1980'den bu yana modern fungisitlerin yağmur dayanıklılığında büyük bir gelişme olmuştur. Aslında en modern fungisitlerin yaprak üzerinde kuruma şansları varsa 30 mm'ye kadar yağmura dayanmalarını bekleyebiliriz. Eğer yağmur ilaçlamadan hemen sonra ya da ilaçlama sırasında başlarsa yağmur direnci büyük ölçüde azalabilir.
Kontakt fungisitlerin eski moda formülasyonlarında 12 mm'den daha az bir yağmur sertliği beklememiz gerekir. Tıpkı 1970'lerde alışık olduğumuz gibi. Bir bağdaki yaprakları ıslatmak için yaklaşık 2 mm yağmura ihtiyaç vardır. Bu nedenle bu modülde, bir yaprak ıslatma dönemi içinde sadece 2 mm'den büyük yağmurları biriktiriyoruz. Bu, tek bir gün içinde toplam 6 mm yağmur olabileceği anlamına gelir, ancak bu modül bunların hiçbirini biriktirmez çünkü yapraklar 2 mm yağmur yağmadan önce tekrar kurumuştur.
Yağmur 3, 5 ve 7 gün boyunca biriktirilir. Daha uzun bir süre boyunca bitki büyümesi, temas fungisitlerinin etkisi için bileşiklerin yağmur direncinden çok daha önemlidir.
Ateş yanıklığı
Kaynak:P.W. Steiner, T. van der Zwet ve A. R. Biggs
Ateş yanıklığı, elma ve armutlarda çiçekleri, sürgünleri, dalları ve bazen de tüm ağaçları öldüren yıkıcı bir bakteriyel hastalıktır. Hastalık dünya çapında neredeyse tüm orta ve sıcak elma yetiştirme alanlarında görülür. Salgınlar tipik olarak çok düzensiz olmasına rağmen, bazı yıllarda bazı bahçelerde ciddi kayıplara neden olurken, diğerlerinde çok az veya hiç önemli hasara neden olmaz. Bu düzensiz oluşum, kışlayan inokulumun mevcudiyetindeki farklılıklara, enfeksiyonu yöneten özel gereksinimlere, belirli yerel hava koşullarındaki değişikliklere ve mevcut çeşitlerin gelişim aşamasına bağlanmaktadır. Ateş yanıklığının yıkıcı potansiyeli ve düzensiz doğası, salgınların genellikle birkaç farklı aşamada geliştiği gerçeğiyle birlikte, bu hastalığın kontrolünü zor ve maliyetli hale getirir.
Semptomlar
Ateş yanıklığı patojenini barındıran kışı geçiren çürükler genellikle gövdelerde ve büyük dallarda, bazen kenarları çatlamış, rengi bozulmuş kabuğun hafif ila derin çöküntü alanları olarak açıkça görülebilir. Bununla birlikte, en büyük sayıdaki çürükler çok daha küçüktür ve kolayca ayırt edilemez. Bunlar, bir önceki yıl çiçek veya sürgün enfeksiyonlarının meydana geldiği küçük dallarda ve genellikle yanık dalları çıkarmak için yapılan kesiklerin etrafında görülür. Bu çürüklerin çoğu mevsimin ilerleyen zamanlarında oluştuğu için, genellikle güçlü bir şekilde bastırılmazlar ve nadiren kenarlarında kabuk çatlakları görülür. Ayrıca, genellikle oldukça küçüktürler, 2,5 cm'den daha az uzanırlar, kırmızımsı ila mor kabukları küçük siyah mantar meyve gövdeleriyle kaplı olabilir (en önemlisi Botryosphaeria obtusaelmanın kara çürüklük patojeni).
Çiçek yanıklığı belirtileri çoğunlukla çiçeklenmeden sonraki bir ila iki hafta içinde ortaya çıkar ve genellikle solup ölen, elmada kahverengiye ve armutta oldukça siyaha dönen tüm çiçek kümesini içerir. Hava patojen gelişimi için uygun olduğunda, çiçeklerin üzerinde bakteriyel sızıntı kürecikleri görülebilir. Çiçek salkımını taşıyan mahmuz da ölür ve enfeksiyon destekleyici uzvun bazı kısımlarına yayılabilir ve onları öldürebilir. Enfekte olmuş genç sürgünlerin uçları solgunlaşarak çok tipik bir "çoban kavuğu" belirtisi oluşturur. Yaklaşık 20 yaprak geliştirdikten sonra enfekte olan daha yaşlı sürgünler uçta bu kıvrılma belirtisini göstermeyebilir. Enfeksiyon sürgün ekseninde yayıldıkça, yapraklar önce orta damarlarda koyu çizgiler gösterir, ardından solgunlaşır ve kahverengiye döner ve sezon boyunca sürgüne sıkıca bağlı kalır. Çiçek enfeksiyonlarında olduğu gibi, patojen genellikle enfekte sürgünü destekleyen uzvun bir kısmını istila eder ve öldürür. Yakındaki aktif çürüklüklerden sistemik olarak istila edilen su filizleri ve sürgünlerdeki ilk belirti, solma meydana gelmeden önce sürgün ucunda sarı ila turuncu renk değişiminin gelişmesidir. Buna ek olarak, bu tür filizlerin taban yapraklarının yaprak sapları ve orta damarları genellikle sürgün ucundakilerden önce nekrotik hale gelir.
Çeşitlere ve enfeksiyonun meydana geldiği zamanki gelişim aşamasına bağlı olarak, tek bir çiçek veya sürgün enfeksiyonu tüm bir uzvun ölümüyle sonuçlanabilir ve ağacın merkezi lideri veya gövdesi istila edildiğinde, ağacın büyük bir kısmı sadece bir sezonda öldürülebilir. Genel olarak, taç yaprak dökümü ile terminal tomurcuk oluşumu arasında meydana gelen her tür enfeksiyon genellikle en büyük uzuv ve ağaç kaybına yol açar. Buna ek olarak, ağır yapılı ağaçlar, yüksek verimlilik için daha zayıf sistemlerle eğitilenlere göre daha az ciddi uzuv kaybına uğrama eğilimindedir. Oldukça duyarlı elma anaçlarının (M.26, M.9) enfekte olduğu durumlarda, aşı birliğinin üzerindeki aşı gövdesi ve ana dalların çoğu tipik olarak belirtisiz kalırken, anaç çevresinde belirgin koyu kahverengi bir çürüklük gelişir. Bu anaç çürüklüğü ağacı çevreledikçe, üst kısım sezon ortasından sonuna kadar genel düşüş belirtileri (zayıf yaprak rengi, zayıf büyüme) gösterir. Bazı durumlarda, anaç yanıklığından etkilenen ağaçların yaprakları, toprak kaynaklı bir mantarın neden olduğu yaka çürüklüğü hastalığıyla ilişkilendirilenden farklı olarak, Ağustos sonu ile Eylül başında erken sonbahar kırmızı rengi geliştirir. Anaç enfeksiyonu olan bazı ağaçlar bir sonraki bahara kadar gerileme belirtileri göstermeyebilir ve bu dönemde alt gövdeye doğru uzanan çürüklükler görülebilir.
Hastalık Döngüsü
Ateş yanıklığına neden olan bakteriyel patojen neredeyse sadece bir önceki sezonda enfekte olmuş dallardaki kan çukurlarında kışı geçirir. En fazla sayıda kanber ve dolayısıyla inokulum katkısında en önemli olanlar, çapı 38 mm'den küçük dallarda, özellikle de bir önceki yıl yanık dalları çıkarmak için yapılan kesiklerin etrafında meydana gelir. İlkbaharın erken dönemlerinde daha yüksek sıcaklıklar ve hızlı tomurcuk gelişimiCanker kenarlarındaki bakteriler hızla çoğalmaya başlar ve çiçeklenme döneminden birkaç hafta öncesine kadar kabuk yüzeyinde detaylandırılan kalın sarımsı ila beyaz bir balçık üretir. Birçok böcek türü (çoğunlukla sinekler) bu balçığa ilgi duyar ve ardından bakterileri meyve bahçesi boyunca dağıtır. İlk birkaç açık çiçek bakteri tarafından kolonize edildiğinde, tozlaşan böcekler patojeni hızla diğer çiçeklere taşıyarak daha fazla çiçek yanıklığı başlatır. Kolonize olan bu çiçekler, herhangi bir çiçek yanıklığı vakasından birkaç dakika sonra enfeksiyona maruz kalır. ıslanma olayı neden olduğu yağmur veya yoğun çiy ne zaman ortalama günlük sıcaklıkların 16 °C'ye eşit veya daha yüksek olması Çiçek taç yaprakları sağlamken (çiçek kapları ve genç meyveler taç yaprakları düştükten sonra dayanıklıdır). Çiçek enfeksiyonları ortaya çıktıktan sonra, erken belirtiler, çiçek taç yapraklarında 13 °C'den yüksek en az 57 derece gün (DD) Bu da günlük sıcaklıklara bağlı olarak 5 ila 30 takvim günü gerektirebilir.
Çiçek yanıklığı belirtilerinin ortaya çıkmasıyla birlikte, çiçek yanıklığının sayısı ve dağılımı aşı kaynakları meyve bahçesinde büyük ölçüde artar. Bu kaynaklardan gelen inokulum şu yollarla daha da yayılır rüzgar, yağmur ve birçok sıradan böcek ziyaretçisi genç sürgün uçlarına ulaşarak sürgün yanıklığı salgını olasılığını artırır. Pennsylvania'da yapılan son araştırmalar yaprak biti beslenmesinin sürgün yanıklığına katkıda bulunmadığını göstermektedir. Yaprak zararlılarının sürgün yanıklığı insidansında rol oynayıp oynamadığını belirlemek için daha fazla araştırmaya ihtiyaç vardır. Sürgün ucu enfeksiyonlarının çoğu, sürgünlerin yaklaşık dokuz ila on yapraklı olduğu zaman ile terminal tomurcuk seti arasında, inokulum kaynaklarının ve böcek vektörlerinin mevcut olduğu ve günlük sıcaklıkların ortalama 16 ° C veya daha fazla olduğu zamanlarda meydana gelir.
Çiçek enfeksiyonlarının görülmediği yıllarda birincil inokulum kaynakları sürgün yanıklığı aşaması için kışlayan kan çıbanları ve özellikle de bu çürüklüklerin yakınındaki genç su filizleri, bakterilerin çürüklük kenarlarından sistemik olarak bu filizlere geçmesiyle enfekte olur. Canker yanıklığı olarak adlandırılan bu tür sistemik sürgün enfeksiyonları, yeşil uçtan sonra 13 °C'den yaklaşık 111 DD daha büyük bir sürede başlar, ancak görünür semptomlar yeşil uçtan sonra 13 °C'den en az 157 DD daha büyük bir birikime kadar belirgin olmayabilir. Çiçek enfeksiyonlarının yokluğunda, sürgün yanıklığı enfeksiyonlarının gelişimi genellikle kışı geçiren çürüklüklerin bulunduğu alanların etrafında lokalize olur.
Olgun sürgün ve uzuv dokuları genellikle enfeksiyona karşı dirençli olmasına rağmen E. amylovoradolu, -2 °C veya daha düşük geç donlar ve yapraklara zarar veren şiddetli rüzgarların neden olduğu yaralanmalar, olgun dokulardaki normal savunma mekanizmalarının ihlal edildiği ve enfeksiyonların meydana geldiği bir travma yanıklığı durumu yaratabilir. Travma yanıklığı vakalarının 'Delicious' gibi normalde dayanıklı çeşitlerde bile meydana geldiği bilinmektedir.
Ateş yanıklığının bir başka aşaması olan anaç yanıklığı son zamanlarda tanınmıştır ve öncelikle oldukça hassas M.26, M.9 ve Mark anaçları ile ilişkilidir. Bu ağaçlarda, aşı çeşidindeki sadece birkaç çiçek veya sürgün enfeksiyonu, daha sonra sistemik olarak anaca taşınan bakterileri sağlayabilir ve burada her zaman olmasa da genellikle bir çürüklük gelişir ve sonunda ağacı çevreler. Anaç yanıklığından etkilenen ağaçlar genellikle mevsimin ortasından sonuna kadar gerileme ve erken ölüm belirtileri gösterir, ancak bir sonraki bahara kadar belirgin olmayabilir.
(c) Dr. Heinrich Denzer, Pessl Instruments GmbH, Weiz, 2007
Armutlar için Model Puma Yanıklığı
Semptomlar
Model, kullanıcının aşağıdakileri tanımasını gerektirir özel ve sürekli değişen yerel etkinlikler ve bahçelerinin bölgedeki diğer meyve bahçelerine göre yangın yanıklığı riskini artırabilecek veya azaltabilecek yönleri. Model, kullanıcının, ağaçlarda çiçeklerin bulunduğu her zaman, özellikle de aşağıdaki dönemlerde yangın yanıklığı enfeksiyonu riski olduğunu varsaymasını gerektirir taç yaprağı dökümü ve "çiçeklenme sonrası" dönem, Birçok elma ve armut çeşidinde dağınık çiçeklerin kalabileceği zaman. Model kullanıcısından kendi sahasındaki durumu dikkatlice değerlendirmesi ve çiçeklerin mevcut olması, risk seviyelerinin "Yüksek" veya "Aşırı" olması ve önümüzdeki 24 saat içinde çiçek ıslanmasının meydana gelme olasılığının yüksek olması durumunda kontrol önlemlerini başlatması istenmektedir.
Model Yapısı
Sıcaklıklar ve Islaklık: Modellenmesi gereken temel Ateş Yanıklığı süreci, elma ve armut çiçeklerinin stigmalarında bakteriyel büyüme potansiyelidir. Bu büyüme sıcaklığa bağlıdır, bu nedenle enfeksiyon riskinin güvenilir bir şekilde tahmin edilmesi, Erwinia amylovora kolonilerinin büyümesini en doğru şekilde yansıtan bir ölçüm yönteminin kullanılmasını gerektirir. Modeller arasındaki temel anlaşmazlık bunun nasıl yapılması gerektiğidir.
FieldClimate'de Yangın Yanıklığı Modeli Çıktısı
Cougarblight modeli, belirli bir büyüme oranı eğrisine dayalı olarak derece saatleri ile bakteriyel büyüme oranını tahmin eder. Bu büyüme eğrisi aşağıdakilerin büyüme oranına dayanmaktadır E. amylovora laboratuvar testlerinde bakteriler. Derece saat değerleri, sıcaklıkların 15 °C'nin üzerinde olduğu günün her saatinde biriktirilir. Saatlik değerler, sıcaklıklar 15°C'den 29°C'ye yükseldikçe artar, daha yüksek sıcaklıklarda düşer ve 40°C'nin üzerindeki sıcaklıklarda bir saat boyunca sıfıra ulaşır.
Armutlar için Model Çiçek Yanıklığı
- Çiçek, stigmalar ve taç yapraklar sağlam olacak şekilde açık olmalıdır, kolonizasyon için stigmalar açıkta olmalıdır, taç yaprak dökülmesindeki çiçekler dirençlidir;
- Son 66 °C gün > 4,4 °C içinde en az 110 °C saat > 18,3 °C birikimi, meyve bahçesindeki en eski açık ve dolayısıyla en kolonize çiçek için epifitik enfeksiyon potansiyelini tanımlar
- Çiy veya 0,2 mm yağmur ya da önceki gün 2,5 mm yağmur şeklinde meydana gelen bir ıslanma olayı, bakterilerin kolonize stigmalardan nektartodlara hareket etmesini sağlar
- Günlük ortalama sıcaklık >= 15,6 °C: Bu, bakterilerin nektartodlara göç etme hızını ve enfeksiyon oluşturmak için gereken bakteri çoğalmasını etkileyebilir.
Bu minimum gerekliliklerin dördü de gösterilen sırayla karşılandığında, enfeksiyonlar meydana gelir ve çiçek yanıklığının ilk erken belirtilerinin ek bir 57 °C gün > 12,7 °C birikimi ile ortaya çıkması beklenebilir. Bu, enfeksiyondan 5 ila 30 gün sonra olabilir. Meyve bahçesi koşulları bu minimum gerekliliklerin altında olduğunda, çok az belirti ortaya çıkar veya hiç belirti görülmez ve önemli bir salgın gelişmez. (STEINER P.W. 1996)
Grafiksel Sunum Armutlarda Yangın Yanıklığı
FieldClimate'de iki ateş yanığı modeli tek bir grafikte gösterilir. Cougar Blight modeli Fire Blight DIV ve Maryblight modeli Blossomblight olarak adlandırılmıştır. Cougar Yanıklığı sonuçlarını yorumlamak için grafiğin altı 5 farklı renkle çizilmiştir. Bu renklerin dağılımı, bahçenin ilk yanıklık geçmişindeki ayarlar temel alınarak yapılmıştır. Bu 5 renk DIV değerleri için risk sınıfını göstermektedir.
Bir Çiçek Enfeksiyonu fırsatı, aynı grafikte 0 - 1 arasında değişen bir çubukla gösterilir (koşullar yerine getirilir veya getirilmez). Meyve bahçesinin geçmişiyle ilgili ayarlar bu modele entegre edilmemiştir. FieldClimate.com'da çiçek enfeksiyonu içeren bir çubuk her hesaplandığında ateş yanığı enfeksiyonu söz konusudur!
Pratik Kullanım Armutlarda Yangın Yanıklığı
Yangın Yanıklığı Modellerinin amacı, aşağıdakiler tarafından enfeksiyon olasılığını değerlendirmektir Erwinia amyloflora meyve bahçesinde.
Bu Mary Blight modeli çiçek yanıklığı için değerlendirilen, yüksek ekonomik etkiye sahip enfeksiyon durumlarını çok iyi gösterir. Bu nedenle bu patojene karşı antibiyotik kullanımını belirtmek için oldukça sık kullanılır.
Puma Yanıklığı patojenin genel yayılma olanaklarına bağlı olarak ateş yanıklığı enfeksiyonu riski hakkında bilgi vermektedir. Bir meyve bahçesinin geçmişine göre yapılan ağırlıklandırma, Mary Blight'ın bir enfeksiyon belirtisi göstermeyeceği durumlarda bile meyve bahçelerini ateş yanıklığı belirtileri açısından ne kadar dikkatli kontrol etmemiz gerektiğini bize göstermesi açısından çok faydalıdır.
(c) Dr. Heinrich Denzer, Pessl Instruments GmbH, Weiz, 2008
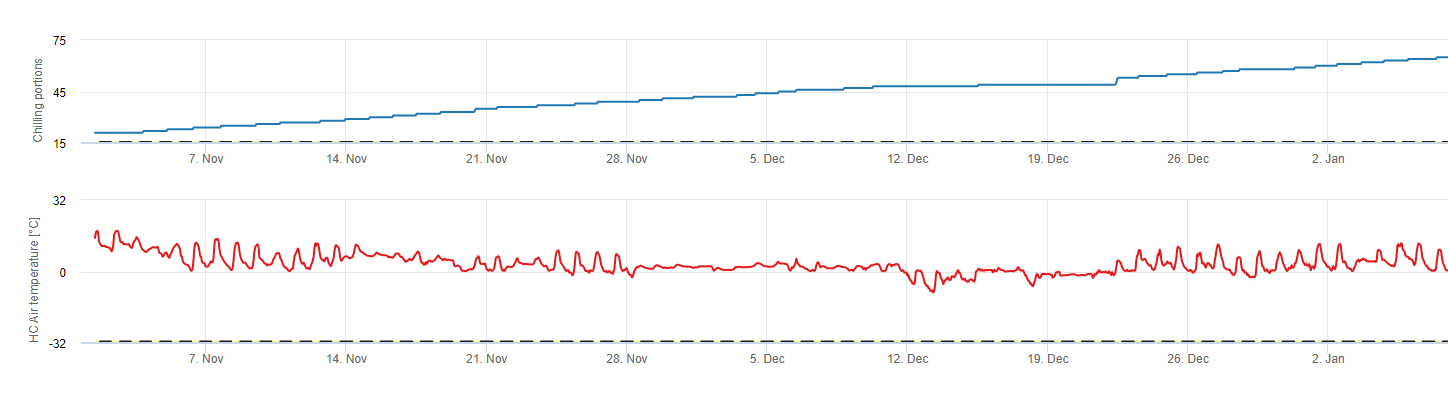
Soğutucu porsiyonlar
Chilling
Sert çekirdekli meyve ağaçları vejetatif ve meyve veren tomurcuklarını yazın geliştirir ve kış yaklaştıkça, zaten gelişmiş olan tomurcuklar hem daha kısa gün uzunluklarına hem de daha düşük sıcaklıklara tepki olarak uykuya geçer. Bu uyku hali ya da uyku aşaması, bu tomurcukları yaklaşan soğuk havadan korur. Tomurcuklar uyku haline girdikten sonra, donma noktasının çok altındaki sıcaklıklara karşı toleranslı olacak ve kış ortasındaki sıcak dönemlere tepki olarak büyümeyeceklerdir. Bu tomurcuklar yeterli soğuk hava birimi (CU) biriktirene kadar uykuda kalırlar. Ne zaman yeterince soğuk biriktiğinde, tomurcuklar ılık sıcaklıklara tepki olarak büyümeye hazırdır. Yeterli CU olduğu sürece çiçek ve yaprak tomurcukları normal şekilde gelişir. Eğer tomurcuklar kış aylarında yeterli soğutma sıcaklığı almazlar ağaçlar yetersiz soğuklama ile ilişkili fizyolojik semptomlardan bir veya daha fazlasını geliştirecektir: 1) yapraklanmanın gecikmesi, 2) meyve tutumunun azalması ve iliklenmenin artması ve 3) meyve kalitesinin düşmesi.
Yetersiz Üşüme Belirtileri
Gecikmiş Yapraklanma:
Yetersiz soğutmanın klasik bir belirtisi yapraklanmanın gecikmesidir. Bir ağaçta gövde uçlarına yakın küçük bir tutam yaprak olabilir ve uçların 12 ila 20 inç altında yaprak bulunmayabilir. Alt tomurcuklar sonunda kırılır ancak tam yapraklanma önemli ölçüde gecikir, meyve tutumu azalır ve ağaç zayıflar. Ayrıca, ağacın alt kısımlarından yoğun emilim yönetim sorunlarına neden olur ve gelecek yılın meyve tomurcuklarının normal gelişimi bozulabilir.
Azaltılmış Meyve Tutumu ve Düğmeleme:
Yetersiz soğuklamaya tepki olarak çiçeklenme, genellikle yaprak gelişiminde görülen modeli takip eder. Çiçeklenme gecikir, uzar ve pistil ve polen gelişimindeki anormallikler nedeniyle meyve tutumu azalır. Birçok şeftali çeşidinde çiçekler kabuk yarılmasından önce veya kabuk yarılması civarında dökülür, ancak 'Jersey Queen' ve 'Harvester' gibi diğerlerinde düğmeler oluşur. Düğmeler, görünüşte açan ama asla tam boy meyveye dönüşmeyen çiçeklerden kaynaklanır. Meyveler olgunlaştıkça küçük ve şekilsiz kalır. Eğer bu meyveleri kesip açarsanız, tohum ölür. Düğümlenme sezonun başlarında belirgin olmadığından, yetiştiriciler anormal meyveleri seyreltemez ve gelişen düğmeler böcekler ve hastalıklar için bir besin kaynağı ve kışlama alanı olarak hizmet eder.
Meyve Kalitesinde Azalma:
Yetersiz soğuklamanın meyve kalitesi üzerindeki etkileri muhtemelen en az tartışılan konudur ancak özellikle orta ve güney Teksas'ta çok yaygın olduğu görülmektedir. Yaprak büyümesi ve meyve tutumu üzerindeki etkiler dramatiktir ancak yetersiz soğuklamanın meyve kalitesi üzerindeki etkileri hafiftir ve diğer belirtiler görülmediğinde ortaya çıkabilir. Yetersiz soğuklama birçok çeşidin ucunun genişlemesine ve sertliğinin azalmasına neden olur. Ayrıca, meyve zemin rengi normalden daha yeşil olabilir, bunun nedeni muhtemelen zemin rengi yeşilden sarıya tam olarak değişmeden önce meyvenin sertliğini kaybetmesidir. Bu kalite sorunlarının kapsamı çeşide ve soğuklama eksikliğinin derecesine bağlıdır.
Modeller
Soğutmayı hesaplamak için kullanılan ve her biri soğutma biriminin ne olduğunu tanımlayan çeşitli modeller vardır. En yaygın üç model 45 derece F (7°C) altındaki saat sayısı modeli, 32 ile 45 derece F (2 ile 7°C) arasındaki saat sayısı modeli ve Utah modelidir. İlk iki model basittir ve bir soğutma birimini belirli sıcaklıkların altında veya arasında bir saat olarak tanımlar. Utah yöntemi daha karmaşıktır çünkü göreceli soğutma etkinliği ve negatif soğutma birikimi (veya soğutma olumsuzlaması) kavramlarını ortaya koymaktadır.
FieldClimate'de şunları kullanırız soğuk porsiyonların hesaplanması için model (CP). Soğutma birikimleri, 2 ila 7°C'lik bir sıcaklık aralığı kullanılarak soğutma kısımları olarak hesaplanır. Soğutma oranlarının hesaplanması 96 saatlik eşit veya daha fazla sürenin ardından >15°C' de sona erer (7 ila 15°C arasında tutar)
Hesaplamalar Erez A, Fishman S, Linsley- Noakes GC, Allan P (1990) Şeftali tomurcuklarında dinlenme tamamlanması için dinamik model çalışmasına dayanmaktadır. Acta Hortic 276: 165-174.
Armut kabuğu
Kaynak: J.W. Travis, J.L. Rytter ve K.S. Yoder
Giriş
Armut kabuklubiti dünya çapında ekonomik açıdan önemli bir hastalıktır ve duyarlı çeşitlerde ciddi kayıplara neden olabilir. Hastalık Kuzey Amerika'ya kıyasla Avrupa ülkelerinde daha büyük bir sorundur ve özellikle Japonya'da büyük bir endişe kaynağıdır. Bazen kara leke olarak da adlandırılan armut kabuğu, elma kabuğuna benzer (Venturia inaequalis) ile neredeyse her açıdan benzerlik gösterir ve yakından ilişkili bir mantardan kaynaklanır, V. pirina. Armut çeşitleri kabuklanmaya karşı duyarlılık bakımından farklılık gösterir; ancak ülkenin bir bölgesinde dayanıklı olan çeşitler başka bir bölgede dayanıklı olmayabilir.
Semptomlar
Armut kabuğunun belirtileri elma kabuğuna çok benzer. Yapraklar ve yaprak sapları üzerindeki lezyonlar yuvarlak, kahverengimsi lekeler olarak başlar ve sonunda kadifemsi bir görünüm alır. Bu lezyonların içinde konidiler üretilir. Sezonun ilerleyen dönemlerinde yaprakların alt yüzeyinde küçük lekeler görülebilir. Bunlar genellikle geç ilkbahar veya erken yaz enfeksiyonlarının sonucudur. Armutta yaprak enfeksiyonu elma yapraklarındaki elma kabuklubiti kadar yaygın değildir.
Hastalık Döngüsü
Meyvelerdeki kabuk lezyonları kaliks ucunda ve sonunda meyvenin yanlarında meydana gelir. Bu lezyonlar büyüdükçe koyu kahverengiye dönüşür ve birleştikçe büyük siyah alanlar oluşturur. Olgunlaşmamış meyvelerdeki lezyonlar küçük, dairesel, kadifemsi lekelerdir. Meyve olgunlaştıkça daha koyu, noktasal lekeler gelişir. Enfekte meyve genellikle düzensiz şekilli olur. Elma kabuklubitinin aksine, armut kabuklubitinde dal enfeksiyonları yaygındır. Büyüme mevsiminin başlarında, genç sürgünlerdeki lezyonlar kahverengi, kadifemsi lekeler olarak görülür. Daha sonra bu lezyonlar mantarımsı, çıban benzeri alanlar haline gelir. Bir sonraki ilkbaharda, kışı geçiren bu lezyonların içinde püstüller gelişecektir. Bu püstüller, hastalığın yayılmasını sürdüren sporlar (konidya) üretir. Mantar kışı yerdeki yapraklarda ve ayrıca enfekte dallarda miselyum olarak geçirir. Armut yapraklarının ve meyvesinin enfeksiyonu, elma kabuk mantarı tarafından elmanın enfeksiyonu için gerekli olanlara benzer koşullar altında gerçekleşir. Askosporlar birincil inokulumun ana kaynağıdır. Enfeksiyon ilkbaharda çiçek tomurcuğu gelişiminin yeşil uç aşaması civarında meydana gelir. Kışı geçiren yapraklardaki askosporlar yağmur sonucu serbest kalır ve hava akımları ile genç yapraklara ve meyvelere taşınır. Askosporlar altı ila sekiz haftalık bir süre boyunca olgunlaşmaya devam eder. Konidiler ikincil inokulumun kaynağıdır ve askosporlar tarafından başlatılan birincil lezyonlarda ya da enfekte dallardaki püstüller içinde üretilir. Bir büyüme sezonu boyunca birçok ikincil döngü meydana gelebilir. Enfeksiyon için gereken ıslanma süresinin uzunluğu ve sıcaklık, sürekli ıslaklık saatlerinin sayısına ve bu ıslanma süresindeki sıcaklığa bağlıdır===. Elma kabuklubiti enfeksiyon sürelerini belirlemek için Mills tablosu, bir yaprak ıslaklık kaydedici veya higrotermograf ile birlikte armut kabuklubiti enfeksiyon sürelerini belirlemek için bilgi sağlayabilir. Kabuk lezyonları genç yapraklarda enfeksiyondan sonra sekiz gün gibi kısa bir sürede, yaşlı yapraklarda ise iki ay gibi uzun bir sürede gelişebilir. Meyveler de gençken daha hassastır; ancak olgun meyveler, ıslanma süresinin uzunluğu yeterince uzunsa enfekte olabilir.
İzleme
Uyku döneminde yetiştiriciler tarafından izlenmesine gerek yoktur. Ascospore olgunluğunun başlangıcını belirlemek için bölgesel Kooperatif Yayım Servisi personeline danışın. Komşu terk edilmiş veya ticari meyve bahçelerindeki kabuk inokulum durumunun farkında olmak, erken sezon kabuk kontrol kararlarını etkileyebilir. Çiçeklenme öncesi dönemde ve meyve tutumu boyunca, hem taze hem de işlenmiş meyveler için, yaprak ıslaklığının süresini ve ıslak dönem boyunca ortalama sıcaklıkları gözlemleyerek armut kabuğu enfeksiyon dönemlerini belirleyin.
Bu Venturia pirina Spotts, R. A. ve Cervantes, L. A. tarafından tasarlanan Enfeksiyon Modeli 1991
Çevresel Girdi değişkenleri: Sıcaklık, ıslaklık süresi.
Model açıklaması: Spotts ve Cervantes, armut fideleri ile yapılan kontrollü ortam deneylerinin yanı sıra, sıcaklık ve ıslaklık süresinin armut fideleri, yaprakları ve meyvelerindeki konidiyal enfeksiyonlar üzerindeki etkilerine ilişkin tarla içi uzuv torbalama deneylerinden elde edilen verileri sunmaktadır. Ascospore enfeksiyon koşullarını değerlendirmemişlerdir ancak konidial enfeksiyon koşullarına oldukça benzer olmaları gerektiğini ve bu nedenle modellerinin Ascospores tarafından birincil enfeksiyonu tahmin etmek için kullanılabileceğini öne sürmektedirler.
Eylem eşiği: Model geliştiricileri, konidilerin yapraklara bulaşması için gereken minimum ıslaklık süresinin, elmanın "hafif" ve "orta" derecede bulaşması için gereken değerler arasında olduğunu gözlemlemiştir. V. inequalis. Mills tablosuna göre askosporlar. Bu nedenle, armut kabuğu Ascospore veya konidia enfeksiyonu için Mills tablosu kullanılırken, yazarlar daha muhafazakar olmak için "hafif" enfeksiyon için ıslaklık saatlerinin kullanılmasını önermektedir.
(c) Dr. Heinrich Denzer, Pessl Instruments GmbH, Weiz, 2009
Fabraea yaprak lekesi
Mantarın neden olduğu yaprak lekesi Fabrea maculatumyaygın ve yıkıcı bir kırmızı uç hastalığıdır (Photinia fraseri), yenidünya (Eriobotrya japonica), Hindistan alıç (Rhaphiolepis indica), bazı armut çeşitleri (Pyrus sp.) ve gül ailesinin diğer bazı üyelerinde görülür. Bu hastalık peyzaj ve fidanlıklardaki bitkilere en çok serin, yağışlı havalarda ve aktif büyümenin gerçekleştiği dönemlerde zarar verir.
Semptomlar
Genişleyen genç yaprakların hem üst hem de alt yüzeylerindeki küçük, dairesel, parlak kırmızı lekeler Entomosporium yaprak lekesi. Ağır hastalıklı yapraklarda çok sayıda küçük leke birleşerek büyük kestane rengi lekelere dönüşebilir. Olgun yapraklardaki yaprak lekeleri kül kahverengisinden açık griye kadar değişen merkezlere ve koyu kırmızıdan kestane rengine kadar değişen belirgin bir sınıra sahiptir. Mantarın spor üreten organları olan küçük siyah lekeler genellikle her yaprak lekesinin merkezinde görülebilir. Yapraklardakine benzer lekeler, uzun süreli serin ve yağışlı havalarda yaprak saplarında ve hassas gövde büyümesinde gelişebilir.
Düşük seviyelerdeki yaprak lekesi genellikle kozmetik hasardan biraz daha fazlasına neden olur, ancak gelecekteki enfeksiyonlar için spor kaynağı oluşturur. Ancak şiddetli enfeksiyonlar genellikle erken ve ağır yaprak dökümü ile sonuçlanır. Ağır yaprak dökümü kırmızı ucun peyzaj değerini ciddi şekilde azaltır ve bitkinin ölümüne neden olabilir. Bazı Hindistan alıç çeşitleri kırmızı uç kadar ciddi şekilde etkilenir.
Hastalık Döngüsü
Yapraklar ve genç sürgünler üzerindeki lekeler, bitkinin hayatta kalması için önemlidir. Entomosporium yaprak lekesi mantarı. Düşmüş, hastalıklı yapraklar mantar için daha az önemli kaynaklardır. Yazın sıcak dönemleri hariç, kış sonundan yılın büyük bir bölümüne kadar lekelerin merkezindeki mantar sporu üreten yapılardan yağışlı havalarda spor yığınları salınır. Bu sporlar, sıçrayan su ve rüzgarın bir kombinasyonu ile sağlıklı yapraklara yayılır. Yeni yaprak lekesi belirtileri, ıslak bir enfeksiyon döneminden sonraki 10-14 gün içinde ortaya çıkar.nfeksiyon Modeli: Kendine özgü böcek benzeri bir görünüme sahip dört hücreli konidiler, çoğunlukla kışı geçiren yaprak çöplerinden ve bir kısmı da dal çürüklerinden, yağmurlardan veya üstten sulamadan sıçrayan suyla yayılır. Enfeksiyon için ıslanma süreleri 10°C - 25°C sıcaklıklarda 8 ila 12 saat arasında değişebilir. Lezyonlar enfeksiyon döneminin başlangıcından yaklaşık 7 gün sonra görülmeye başlar. Hastalık yaz sonunda rüzgar ve yağmurun konidileri ağaç boyunca dağıtmasıyla hızla ilerleyebilir. Yaprakların ve meyvenin enfeksiyona karşı duyarlılığı olgunlukla birlikte azalmaz. Neredeyse tüm Avrupa kökenli armutlar bu yaprak lekesine karşı hassastır. Model, yağmurla birlikte yaprak ıslaklığı başladığında başlar. Yaprak ıslaklığı 1 saatten daha uzun süre kesintiye uğradığında durur.
(c) Dr. Heinrich Denzer, Pessl Instruments GmbH, Weiz, 2008
Kahverengi leke
Armuttaki kahverengi lekeye patojenik mantar neden olur Stemphylium vesicariumSarımsak, pırasa, soğan ve kuşkonmazda da hastalığa neden olur. Armutta mantar yaprakları, meyveleri ve daha az oranda dalları enfekte eder. Meydana gelen nekroz ve meyve çürümesi, mantarın stoma ve lentisellere nüfuz etmesi ve konukçuyu enfekte bölgedeki hücreleri öldürmeye zorlayarak kahverengi lekelere neden olan kimyasal bileşiklerin üretilmesinden kaynaklanır. Kahverengi lekeler özellikle Güney Avrupa'da ciddi hasara neden olur. Bununla birlikte, hastalık Hollanda ve Belçika'daki baskın armut çeşidi olan Additionally'da da bulunmuştur ve çok hassastır (Montesinos ve ark., 1995a).
Sıcaklık ve ıslaklık süresinin konidiyal enfeksiyon üzerindeki etkisi S. vesicarium armut üzerinde daha önce çalışılmıştır (Montesinos ve ark., 1995b). Buradaki bulgular bir kahverengi leke tahmin sisteminin geliştirilmesine yol açmıştır (Llorente ve ark., 2000). Avrupa'daki iklim koşulları güneyden kuzeye farklılık göstermektedir. Güney Avrupa'da sıcak dönemlerde bile gök gürültülü fırtınalar beklenebilirken, Kuzey Avrupa'da yaz ortasında bile birkaç saat süren serin yağmur dönemleri meydana gelebilir. Tahmin sisteminin yeniden gözden geçirilmesi ve hatta farklı iklim koşulları altında kullanılmak üzere uyarlanması gerektiği açıktır.
Aşağıdakilere yol açan nemli dönemler Stemphylium versicarium Armut üzerindeki enfeksiyonlar bu modele göre çok uzun olmak zorundadır. Bu model Llorente, I., Vilardell, P., Moragrega, C. ve Montesinos, E. tarafından yürütülen çalışmalara ve Hollanda'dan A. Boshuizen, P.F. de Jong ve B. Heijne tarafından yapılan elektronik meteoroloji istasyonlarına uyarlama çalışmalarına dayanmaktadır. Bu nemli dönemler kesintiye uğrayabilir. Kesintinin uzunluğu bağıl neme veya buhar basıncı açığına bağlıdır.
FieldClimate modelinin hesaplanmasında, bağıl nem 75%'ye eşit veya daha yüksek olduğu sürece kesinti sonsuza kadar sürebilir. 65% ile 74% arasında bir bağıl nem ile nemli dönemin bozulması 12 saat sürebilir. Bağıl nem 55% ile 64% arasındaysa, bozulma 9 saat sürebilir. Bağıl nem 45% ile 54% arasındaysa bozulma 8 saat sürebilir. Bağıl nem 35% ile 44% arasındaysa kesinti 6 saat sürebilir. Bağıl nem 34%'nin altındaysa kesinti 4 saat sürebilir.
Hafif, orta ve şiddetli enfeksiyonların ilerlemesini gösteren eğriler, farklı hastalık geçmişine sahip bahçeler için eylem eşikleri olarak kullanılabilir. Meyve üzerinde 1%'den fazla hastalık görülme sıklığı olan bahçelerde, tüm hafif enfeksiyonlarda hastalığın kimyasal kontrolü yapılmalıdır. Meyvelerde 1%'den daha az hastalık insidansı olan bahçelerde, tamamlanmış tüm orta enfeksiyonlarda kimyasal kontrol yapılmalıdır. Ve şimdiye kadar hiç hastalık görülmeyen bahçelerde Stemphylium Şiddetli enfeksiyonlar 100%'ye ulaşacaksa kontrol yöntemlerine başlanmalıdır.
Koşullar:
Sıcaklık: 8°C - 38°C
Yaprak ıslaklığı > 0 veya bağıl nem >90%
Faktör: 600; maks: 60000 (100% enfeksiyonu).
(c) Dr. Heinrich Denzer, Pessl Instruments GmbH, Weiz, 2009
Önerilen ekipman
Bu ürünün potansiyel hastalıklarını izlemek için hangi sensör setinin gerekli olduğunu kontrol edin.